
PD-1 is an immune checkpoint receptor that is mainly expressed on cytotoxic T cells. Immune checkpoint inhibitors, such as anti-programmed cell death protein 1 (PD-1) antibody, were first used in esophageal cancer to treat metastatic or recurrent disease. 9–11 No targeted agents have been developed for esophageal cancer, and this disease has been understudied in clinical trials over the past several decades. Doublet chemotherapy, consisting of fluoropyrimidine and platinum, is recognized as the first-line chemotherapy, 7,8 and taxane or irinotecan monotherapy was used as salvage-line chemotherapy before the development of immune checkpoint inhibitors. 5,6 In addition, there are few treatment options for metastatic or recurrent patients with esophageal cancer. Patients with early-stage esophageal cancer are often asymptomatic, but the majority of patients are diagnosed with advanced disease and have a poor prognosis. 2–5 The key risk factors associated with ESCC are smoking and alcohol consumption, while the key risk factors of esophageal adenocarcinoma are gastroesophageal reflux disease, obesity, and Barrett esophagus. 2 In Western countries, however, the incidence of esophageal adenocarcinoma is higher than that of ESCC. ESCC is the most common subtype, and the frequency of this disease is highest in Eastern Asia and Eastern Africa. 1 The main histological subtypes are esophageal squamous cell carcinoma (ESCC) and esophageal adenocarcinoma. KeywordsĮsophageal adenocarcinoma, esophageal cancer, esophageal squamous cell carcinoma, immune checkpoint inhibitor, nivolumab, PD-1 antibody, pembrolizumab Article:Įsophageal cancer is the seventh most common malignancy and the sixth leading cause of death from cancer worldwide. Clinical trials to evaluate the efficacy of nivolumab in combination with cytotoxic drugs as first-line chemotherapy, in combination with chemoradiotherapy, or for the treatment of perioperative patients with esophageal cancer are ongoing and might contribute to the establishment of new standard treatments. Additionally, the CheckMate 577 trial has shown that nivolumab improves disease-free survival when used as an adjuvant treatment in patients with resectable tumors who received neoadjuvant chemoradiation followed by surgery, and in whom a pathological complete response was not achieved. 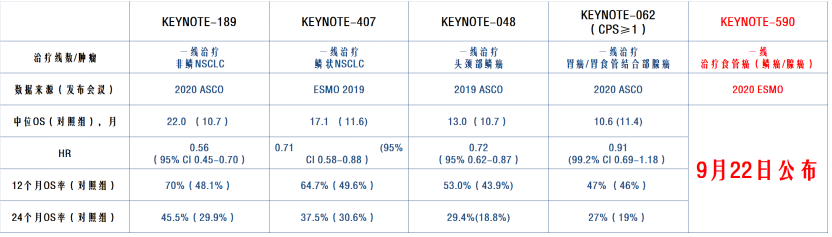
Based on these results, the FDA approved nivolumab as a treatment for patients with advanced ESCC after prior fluoropyrimidine- and platinum-based chemotherapy. The phase III ATTRACTION-3 trial showed the superiority of nivolumab over taxane in patients with advanced esophageal squamous cell carcinoma (ESCC) who were refractory or intolerant to previous fluoropyrimidine- and platinum-based chemotherapy. The phase II ATTRACTION-1 trial showed promising efficacy for nivolumab in patients with advanced esophageal cancer who were refractory or intolerant to fluoropyrimidine-, platinum-, and taxane-based chemotherapies. Nivolumab, a human monoclonal immunoglobulin G4 antibody that inhibits programmed cell death protein 1, has been developed for this patient population. Therefore, the therapeutic development of immune checkpoint inhibitors has been the focus for patients with advanced esophageal cancer. Esophageal cancer is the seventh most common malignancy, for which there are few effective treatment options, especially for patients with advanced disease.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
